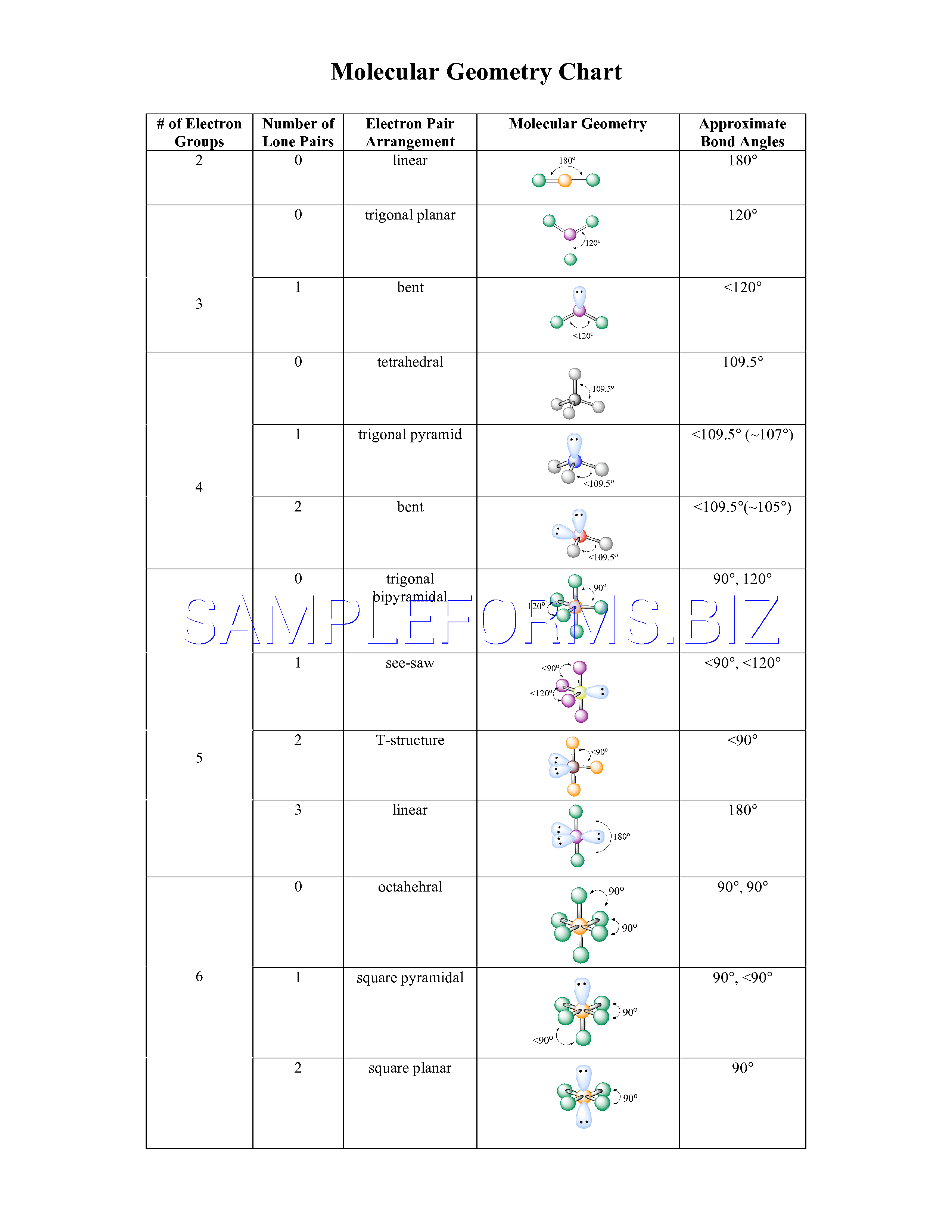 
Bond angles: The angle between adjacent bonds of an atom. Hybridization: Orbitals are combined in order to spread out electrons. Molecular Geometry: Describes the arrangement of atoms around the central atom with acknowledgment to only bonding electrons. The oxygen has two bonding electron pairs (single bond to each H) and two non-bonding pairs giving water a #AX_2E_2# conformation and a bent shape. Electron Geometry: Describes the arrangement of bonds and lone pairs around a central atom. #H_2O# we need to consider the central atom of water which is oxygen. We can use the following notations when examining a Lewis structure of a molecule.Į = non-bonding electron pairs of the central atom 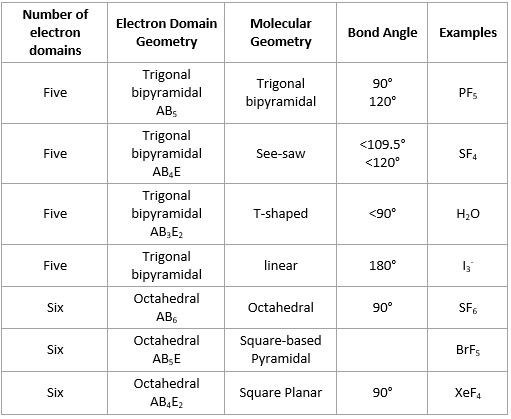
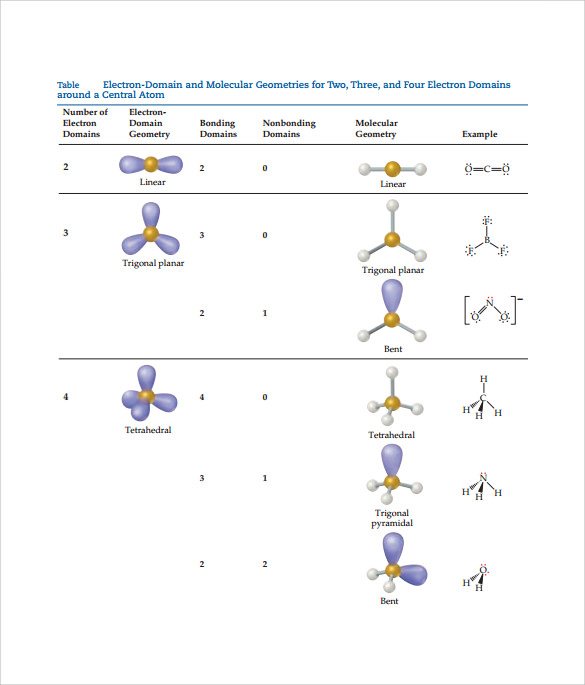
This theory basically says that bonding and non-bonding electron pairs of the central atom in a molecule will repel (push away from) each other in three dimensional space and this gives the molecules their shape. electronic group geometry: trigonal-bi-pyramidal molecular geometry: seesaw ideal angle: 90°, 120° polar, has a dipole moment 4. VESPR stands for valence shell electron pair repulsion. It applies a theory called VESPR for short. Molecular geometry is a way of describing the shapes of molecules. Similar logic applies to all the shapes, you just have to remember which "spoke" will be taken up by an electron pair. Geometry molecular chemistry electron geometries chart shapes bond vsepr notes table science bonding classroom angle group pdf unit teaching basicsGeometry electron molecular In the lewis dot structure for nh3, the central atom of the molecule7.6 molecular structure and polarity chemistry. Once there are any electron pairs, one spoke of the original shape gets "eaten up": for example, a #AX_4E_2# is an octahedron shape, but the two "spokes" are taken up by electron pairs, so you're left with just the square-a square planar shape. #6#: octahedron (a flat square with two "spokes") #5#: trigonal bipyramid (a trigonal planar shape with two "spokes") #3#: trigonal plane (a flat equilateral-triangle-looking shape) As it has a VSEPR shape #AX_5E_0# it is a trigonal bipyramid.Įach steric number has a same "basic shape": Its steric number is #5# due to the #5# bonded atoms to the central #S# atom plus #0# lone electron pairs. The table below summarizes the molecular and electron-pair geometries for different combinations of bonding groups and nonbonding pairs of electrons on the central atom. Thus, it is in the form #AX_3E_1#, which forms a trigonal pyramidal shape. #N#, the central atom, has a steric number of #4#, calculated by the #3# atoms it's bonding with #+1# lone pair. This is the total number of electron pairs and bonds with other atoms. Find the central molecules' steric numbers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
